F2D Medical, French player in innovation for clinical decision-making
Towards predictive and personalised medicine
Innovation • Commitment • Performance • Cohesion
F2D Medical
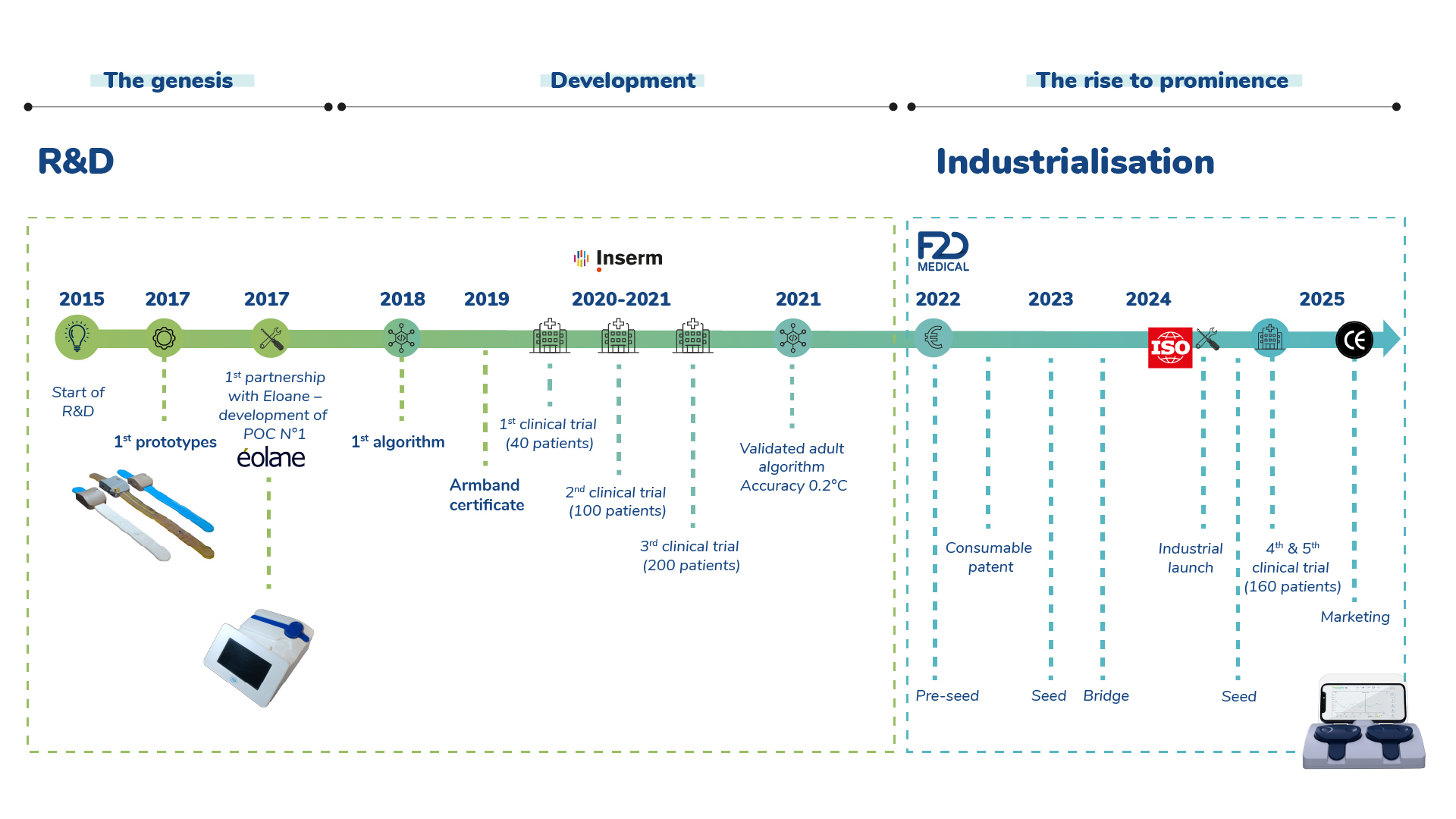
F2D Medical was born from a simple intuition, shared since 2015: why is temperature, an essential factor in diagnosing infections and sepsis, still so unreliable in clinical practice?
Neither contextualised nor standardised, temperature measurement remained untouched by the digital revolution in healthcare.
After five years of R&D, several phases of technical iteration and three clinical studies conducted on 450 subjects, the evidence is clear: a breakthrough is possible.
Thermodiag is now the first non-invasive solution for monitoring core body temperature, designed from the outset for use in the field, integration into the care flow and scientific reliability.
F2D Medical was created in October 2021 with a clear ambition: to transform a strong clinical idea into a certified, secure medical device that is easy to implement and maintain.
Today, F2D Medical is an independent French company, certified to ISO 13485, committed to the development of innovative, useful and accessible medical technologies.
Our mission :
- To put technological innovation at the service of clinical decision-making
- To outpace infectious complications and prevent sepsis
- To relieve healthcare professionals in their daily work with intuitive and interoperable devices
- To contribute to safer, more effective and more humane medicine.


CSR commitment
F2D Medical has chosen to design and manufacture all of its connected medical devices in France. The Normandy-based company has surrounded itself with partners and suppliers who are also ISO 13485 certified and committed to the same CSR approach based on the following principles:
- Ensuring best practices in terms of quality, safety and traceability
- Minimising its environmental impact: reuse, recovery and responsible dismantling
- Supporting medical innovation and connectivity in healthcare facilities
- Promoting the region through sustainable local collaborations
F2D Medical team
At F2D Medical, we share a strong belief: technological innovation is only valuable if it tangibly improves the quality of healthcare and patient journey.
Our team brings together complementary expertise—medical, scientific, technical and operational—to design medical devices with high clinical impact, developed with and for healthcare professionals.
Doctors, biomedical engineers, data scientists, pharmacists, nurses, user experience designers: each of our projects is based on ongoing dialogue with the medical community, rigorous clinical trials and a constant commitment to quality.
An agile structure, a constant commitment
F2D Medical is based on an agile model that promotes rapid iteration, concrete feedback and close relationships with its hospital partners. Our in-house team is supported by a scientific board, a strategic board and an active network of industrial, clinical and technological partners.
We promote a vision of preventive, connected and personalised medicine, providing solutions that are simple to implement and reliable to use — such as Thermodiag, our first device dedicated to the early detection of infections and predictive patient monitoring.

Clinical partners
We would like to thank the many establishments that volunteered to integrate Thermodiag into their clinical trials and pilot projects, helping to advance predictive medicine and the care of at-risk patients.
Groupe Ramsay Santé

Assistance Publique Hôpitaux de Paris
Centre Hospitalier de Bourg-en-Bresse
Centre Unicancer Henri Becquerel Hopital du futur

Hopital Nord Franche-Comté
Centre Hospitalier de Hagueneau

Centre Unicancer Léon Bérard

Centre Hospitalier d’Ajaccio

Our distributors


- FRANCE
- Luquet et Duranton

- ASIA, MIDDLE EAST & AFRICA
- Datavitalis

Industrial, clinical & scientific partners
Eurofins
organisme notifié pour
la certification ISO 13485
et marquage CE
DMH
partenaire clinique
Eurodecision
partenaire data-scientist

Gergonne
partenaire industriel français
certifié ISO 13485

Eolane
partenaire industriel français certifié ISO 13485
Expert boards
Scientific board composed of 7 members:
- Heads of intensive care units
- Oncologist
- Internist
- Pulmonologist
- Emergency doctor
Strategic board:
- Head of intensive care unit
- Pharmacists
- Purchasing manager